Gain more flexibility with outpatient administration
Outpatient administration of VYXEOS was used for some patients in the Phase 3 study, and multiple institutions have evaluated its use in real-world practice.1-4,a
Administration ScheduleThis site is intended for US healthcare professionals only.
Outpatient administration of VYXEOS was used for some patients in the Phase 3 study, and multiple institutions have evaluated its use in real-world practice.1-4,a
Administration Schedule
aMost patients in the Phase 3 study received induction in an inpatient setting.4




Additional considerations for outpatient treatment
In 2 small, postapproval, single-institution studies, more than half of patients initiated VYXEOS induction in the outpatient setting.





In a small, single-center pilot study by Kubal et al, 22 patients received a full induction course of VYXEOS
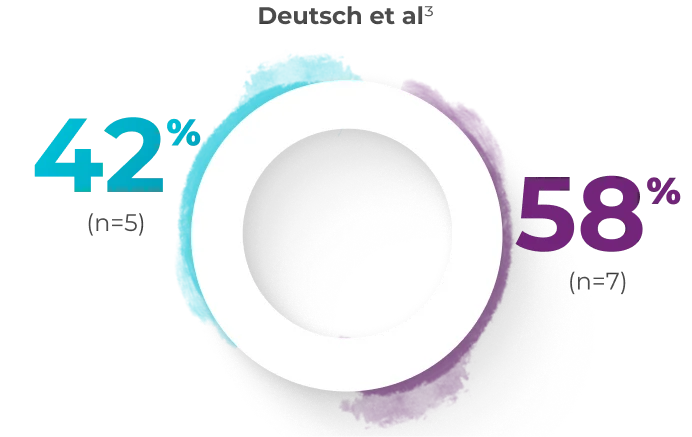
In a small, single-center pilot study by Deutsch et al, 12 patients received a full induction course of VYXEOS
These 2 studies assessed the feasibility of patients receiving VYXEOS induction in the inpatient/outpatient setting2,3
Treatment in an IPOP setting enables appropriate patients to receive induction in an outpatient setting, with inpatient admission scheduled as needed for continued monitoring and care or for treatment for adverse reactions.2,3
ANC=absolute neutrophil count; CBC=complete blood count; CMP=comprehensive metabolic panel; ECOG PS=Eastern Cooperative Oncology Group Performance Status; IPOP=inpatient/outpatient; LDH=lactate dehydrogenase; TLS=tumor lysis syndrome.
bANC >0.5 x 109/L.5
cThe admission stay typically lasts an average of 30 days (ranging between 25 and 50 days).5
dFrequency of assessment may be modified based on patient need.5
Treatment with VYXEOS gives your patients additional flexibility between induction and consolidation cycles.7
more
VYXEOS is prepared for infusion through straightforward steps.7 View helpful guides on how to prepare and administer VYXEOS.
more
VYXEOS is indicated for the treatment of newly-diagnosed therapy-related acute myeloid leukemia (t‑AML) or AML with myelodysplasia-related changes (AML-MRC) in adults and pediatric patients 1 year and older.
WARNING: DO NOT INTERCHANGE WITH OTHER DAUNORUBICIN AND/OR CYTARABINE-CONTAINING PRODUCTS
VYXEOS has different dosage recommendations than daunorubicin hydrochloride injection, cytarabine injection, daunorubicin citrate liposome injection, and cytarabine liposome injection. Verify drug name and dose prior to preparation and administration to avoid dosing errors.
VYXEOS is contraindicated in patients with a history of serious hypersensitivity reactions to cytarabine, daunorubicin, or any component of the formulation.
Serious or fatal hemorrhage events, including fatal CNS hemorrhages, associated with prolonged thrombocytopenia, have occurred with VYXEOS. The overall incidence (grade 1-5) of hemorrhagic events was 74% in the VYXEOS arm and 56% in the control arm. The most frequently reported hemorrhagic event was epistaxis (36% in VYXEOS arm and 18% in control arm). Grade 3 or greater events occurred in 12% of VYXEOS-treated patients and in 8% of patients in the control arm. Fatal treatment-emergent CNS hemorrhage not in the setting of progressive disease occurred in 2% of patients in the VYXEOS arm and in 0.7% of patients in the control arm. Monitor blood counts regularly and administer platelet transfusion support as required.
VYXEOS contains daunorubicin, which has a known risk of cardiotoxicity. This risk may be increased in patients with prior anthracycline therapy, preexisting cardiac disease, previous radiotherapy to the mediastinum, or concomitant use of cardiotoxic drugs. Assess cardiac function prior to VYXEOS treatment and repeat prior to consolidation and as clinically required. Discontinue VYXEOS in patients with impaired cardiac function unless the benefit of initiating or continuing treatment outweighs the risk. VYXEOS is not recommended in patients with cardiac function that is less than normal.
Total cumulative doses of non-liposomal daunorubicin greater than
550 mg/m2 have been associated with an increased incidence of
drug-induced congestive heart failure. The tolerable limit appears
lower
(400 mg/m2) in patients who received radiation therapy to the
mediastinum. Calculate the lifetime cumulative anthracycline
exposure prior to each cycle of VYXEOS. VYXEOS is not recommended in
patients whose lifetime anthracycline exposure has reached the
maximum cumulative limit.
Serious or fatal hypersensitivity reactions, including anaphylactic reactions, have been reported with daunorubicin and cytarabine. Monitor patients for hypersensitivity reactions. If a mild or moderate hypersensitivity reaction occurs, interrupt or slow the rate of infusion with VYXEOS and manage symptoms. If a severe or life-threatening hypersensitivity reaction occurs, discontinue VYXEOS permanently, treat the symptoms, and monitor until symptoms resolve.
VYXEOS contains copper. Consult with a hepatologist and nephrologist with expertise in managing acute copper toxicity in patients with Wilson’s disease treated with VYXEOS. Monitor total serum copper, serum non-ceruloplasmin-bound copper, 24-hour urine copper levels, and serial neuropsychological examinations during VYXEOS treatment in patients with Wilson’s disease or other copper-related metabolic disorders. Use only if the benefits outweigh the risks. Discontinue in patients with signs or symptoms of acute copper toxicity.
Daunorubicin has been associated with severe local tissue necrosis at the site of drug extravasation. Administer VYXEOS by the intravenous route only. Confirm patency of intravenous access before administration. Do not administer by intramuscular or subcutaneous route.
VYXEOS can cause embryo-fetal harm when administered to a pregnant woman. Patients should avoid becoming pregnant while taking VYXEOS. If VYXEOS is used during pregnancy or if the patient becomes pregnant while taking VYXEOS, apprise the patient of the potential risk to a fetus. Advise females and males of reproductive potential to use effective contraception during treatment and for 6 months following the last dose of VYXEOS.
The most common adverse reactions (incidence ≥25%) are hemorrhagic events (74%), febrile neutropenia (70%), rash (56%), edema (55%), nausea (49%), mucositis (48%), diarrhea (48%), constipation (42%), musculoskeletal pain (43%), fatigue (39%), abdominal pain (36%), dyspnea (36%), headache (35%), cough (35%), decreased appetite (33%), arrhythmia (31%), pneumonia (31%), bacteremia (29%), chills (27%), sleep disorders (26%), and vomiting (25%).
Please see full Prescribing Information, including BOXED Warning.
References:
1. Kolitz JE, Strickland SA, Cortes JE, et al. Consolidation outcomes in CPX-351 versus
cytarabine/daunorubicin-treated older patients with high-risk/secondary acute myeloid leukemia. Leuk
Lymphoma. 2020;61(3):631-640.
2. Kubal TE, Salamanca C, Komrokji RS, et al. Safety and feasibility of outpatient
induction chemotherapy with CPX-351 in selected older adult patients with newly diagnosed AML. J Clin
Oncol. 2018;36(15)(suppl):e19013.
3. Deutsch YE, Presutto JT, Brahim A, et al. Safety and feasibility of outpatient liposomal
daunorubicin and cytarabine (Vyxeos) induction and management in patients with secondary AML. Blood.
2018:132(suppl 1):3559.
4. Kolitz JE, Strickland SA, Cortes JE, et al. Efficacy by consolidation administration site: subgroup analysis of a phase 3 study of CPX-351 versus 7+3 in older adults with newly diagnosed, high-risk acute myeloid leukemia (AML). Presented at: American Society of Clinical Oncology Annual Meeting; June 2-6, 2017; Chicago, IL. Poster 7036.
5. Talati C, Frantz D, Lubas A, et al. How I treat newly diagnosed acute myeloid leukemia
in an outpatient setting: a multidisciplinary team perspective. Future Oncol. 2020;16(7):281-291.
6. Aw A, Sabloff M, Sheppard D, et al. Evaluation of an outpatient model for treatment of acute myeloid leukemia. J Hematol. 2016;5(1):1-7.
7. VYXEOS [package insert]. Palo Alto, CA: Jazz Pharmaceuticals.