Safety profile
Comparable safety to 7+3

This site is intended for US healthcare professionals only.

Common adverse reactions (≥20% incidence in the VYXEOS arm) during the induction phase
| Adverse Reaction | All Gradesa | Grades 3 to 5a | ||
|---|---|---|---|---|
| VYXEOS (n=153) n (%) |
7+3 (n=151) n (%) |
VYXEOS (n=153) n (%) |
7+3 (n=151) n (%) |
|
| Hemorrhage | 107 (70) | 74 (49) | 15 (10) | 9 (6) |
| Febrile neutropenia | 104 (68) | 103 (68) | 101 (66) | 102 (68) |
| Rash | 82 (54) | 55 (36) | 8 (5) | 2 (1) |
| Edema | 78 (51) | 90 (60) | 2 (2) | 5 (3) |
| Nausea | 72 (47) | 79 (52) | 1 (1) | 1 (1) |
| Diarrhea/colitis | 69 (45) | 100 (66) | 4 (3) | 10 (7) |
| Mucositis | 67 (44) | 69 (46) | 2 (1) | 7 (5) |
| Constipation | 61 (40) | 57 (38) | 0 | 0 |
| Musculoskeletal pain | 58 (38) | 52 (34) | 5 (3) | 4 (3) |
| Abdominal pain | 51 (33) | 45 (30) | 3 (2) | 3 (2) |
| Cough | 51 (33) | 34 (23) | 0 | 1 (1) |
| Headache | 51 (33) | 36 (24) | 2 (1) | 1 (1) |
| Dyspnea | 49 (32) | 51 (34) | 17 (11) | 15 (10) |
| Fatigue | 49 (32) | 58 (38) | 8 (5) | 8 (5) |
| Arrhythmia | 46 (30) | 41 (27) | 10 (7) | 7 (5) |
| Decreased appetite | 44 (29) | 57 (38) | 2 (1) | 5 (3) |
| Pneumonia (excluding fungal) | 39 (26) | 35 (23) | 30 (20) | 26 (17) |
| Sleep disorders | 38 (25) | 42 (28) | 2 (1) | 1 (1) |
| Bacteremia (excluding sepsis) | 37 (24) | 37 (25) | 35 (23) | 31 (21) |
| Vomiting | 37 (24) | 33 (22) | 0 | 0 |
| Chills | 35 (23) | 38 (25) | 0 | 0 |
| Hypotension | 30 (20) | 32 (21) | 7 (5) | 1 (1) |
| Non-conduction cardiotoxicity | 31 (20) | 27 (18) | 13 (9) | 15 (10) |
Fatal treatment-emergent CNS hemorrhage not in the setting of progressive disease occurred in 2% of patients in the VYXEOS arm and 0.7% in the control arm (7+3).
Other adverse reactions that occurred in ≥10% of patients in the VYXEOS arm included dizziness, fungal infection, hypertension, hypoxia, upper respiratory infections (excluding fungal), chest pain, pyrexia, catheter/device/injection site reaction, delirium, pleural effusion, anxiety, pruritus, sepsis (excluding fungal), hemorrhoids, petechiae, renal insufficiency, transfusion reactions, and visual impairment (except bleeding).
The safety population included all patients in the VYXEOS cohort and 151 patients from the 7+3 cohort (5 patients withdrew consent before the receipt of treatment).1
aAdverse reactions were graded using National Cancer Institute Common Terminology Criteria for Adverse Events version 3.0.
Time to recovery of absolute neutrophil count and platelets may be prolonged with VYXEOS and require additional monitoring2
30- and 60-day overall all-cause mortality in patients with sAML aged 60-75 years (safety population)
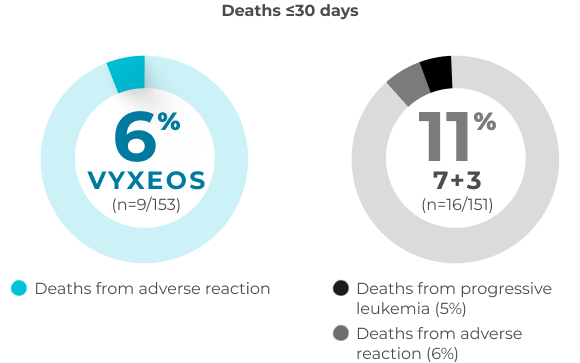

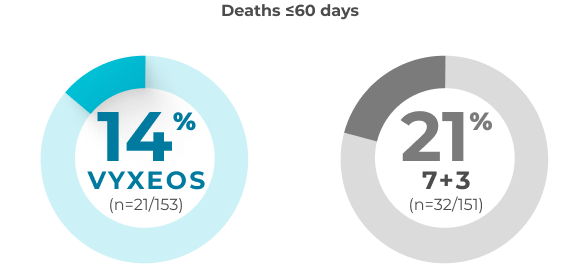
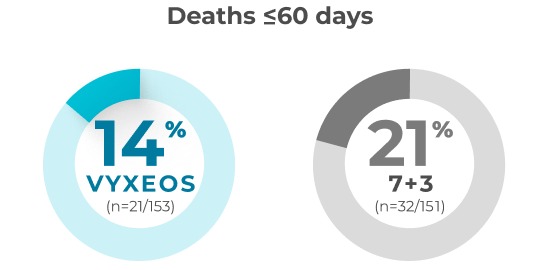
A post hoc analysis was conducted to assess the impact of VYXEOS vs 7+3 on cardiac impairment in a subset of patients from the Phase 3 study.
Percentage of patients with clinically
significantc,d
change in LVEF or GLS3
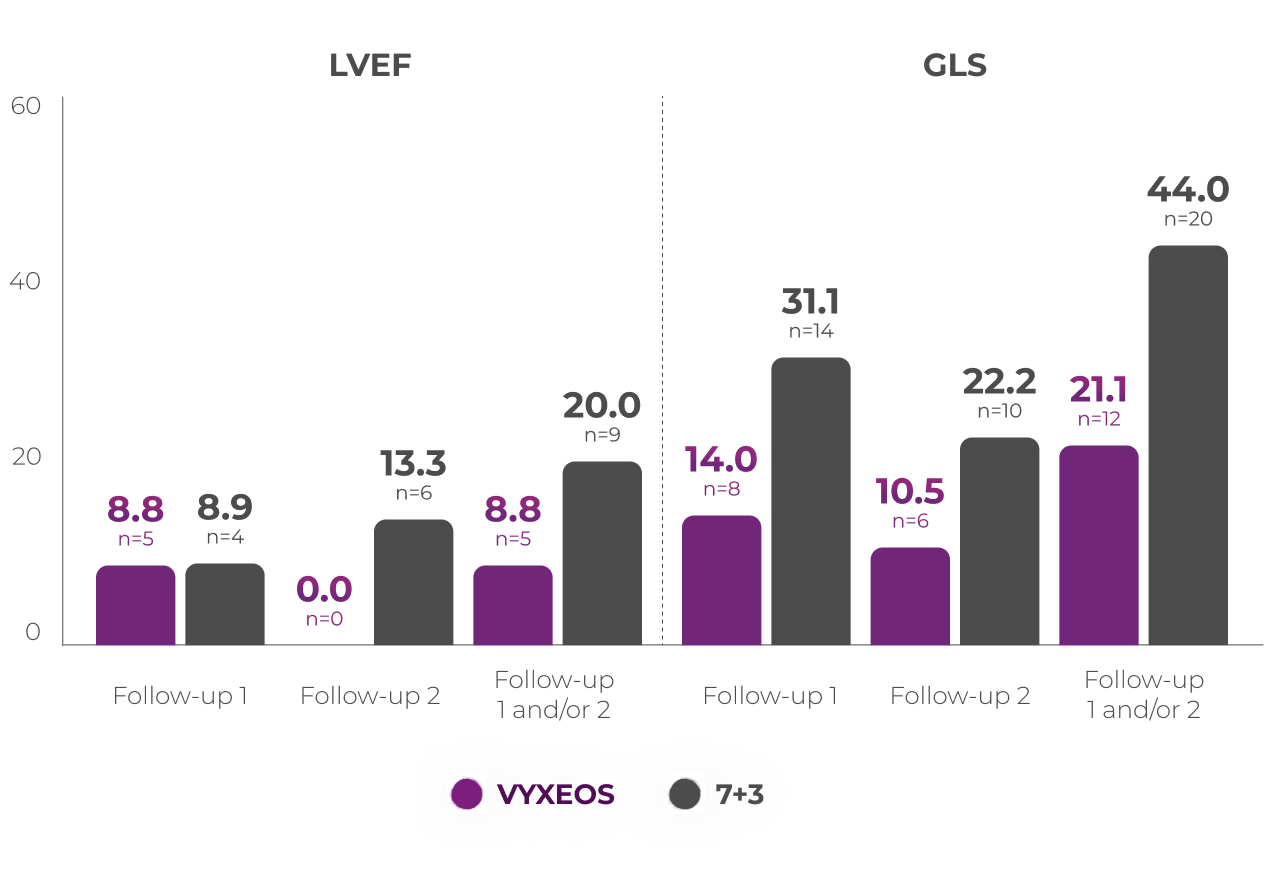
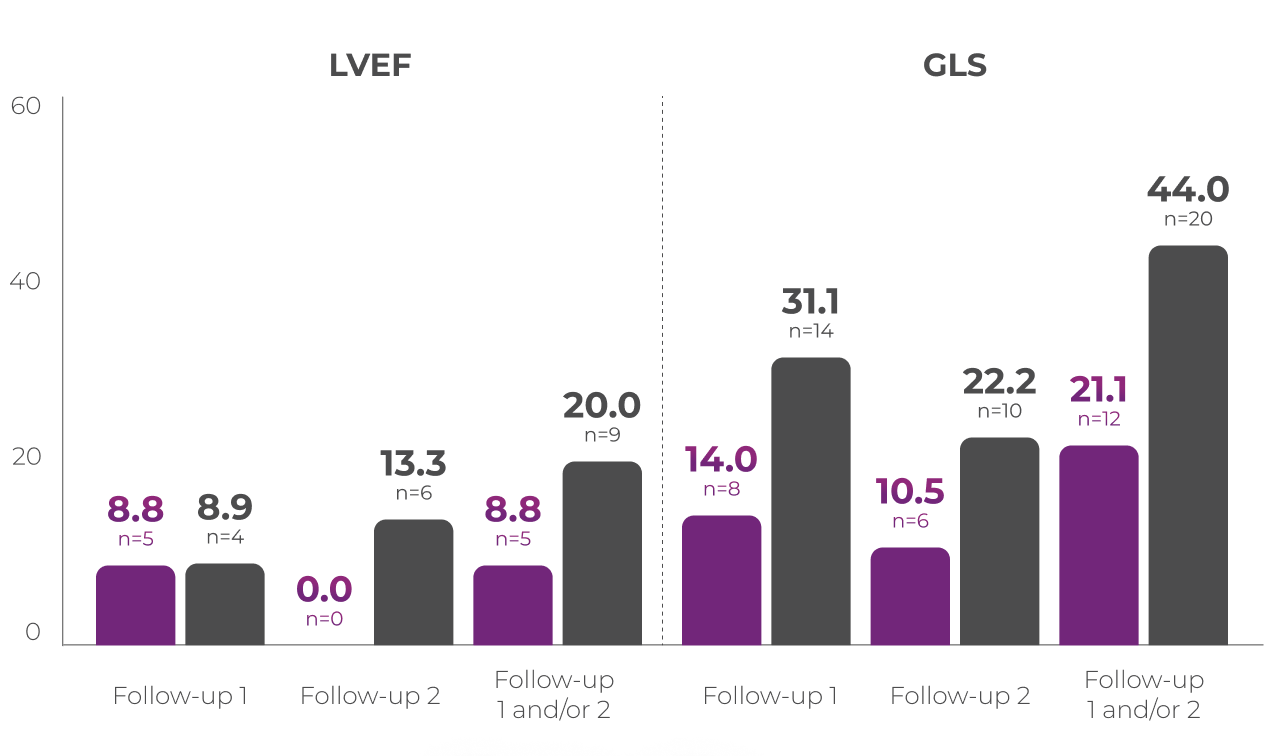

Patients from the Phase 3 study (VYXEOS [n=57]; 7+3 [n=45]) with normal baseline LVEF and at least one postbaseline echocardiogram measure were evaluated. Patients with prior cumulative anthracycline exposure of
>368 mg/m2 daunorubicin (or equivalent) were excluded from the study.3
Cardiac function was assessed at3:
Median baseline LVEFc and GLSd in the VYXEOS and 7+3 arms were3:
Limitations of subanalysis3
The limitations of this analysis include its post hoc design, the restricted patient population, and limited follow-up time points. No conclusions should be drawn. Analysis was not powered to determine statistical significance.
VYXEOS treatment is not recommended in patients with LVEF that is less than normal. Discontinue VYXEOS in patients with impaired cardiac function unless the benefit of initiating or continuing treatment outweighs the risk.2
Some patients may be appropriate for outpatient administration of VYXEOS, giving you more options when planning their treatment. Read how some institutions are approaching outpatient use of VYXEOS.
more
Secondary AML is a broad category of high-risk AML that includes multiple subtypes of leukemias with different characteristics but similar poor prognosis and significant challenge to reach a cure.5,6 Learn more about which patients may be appropriate for intensive chemotherapy with VYXEOS.
more
VYXEOS is indicated for the treatment of newly-diagnosed therapy-related acute myeloid leukemia (t‑AML) or AML with myelodysplasia-related changes (AML-MRC) in adults and pediatric patients 1 year and older.
WARNING: DO NOT INTERCHANGE WITH OTHER DAUNORUBICIN AND/OR CYTARABINE-CONTAINING PRODUCTS
VYXEOS has different dosage recommendations than daunorubicin hydrochloride injection, cytarabine injection, daunorubicin citrate liposome injection, and cytarabine liposome injection. Verify drug name and dose prior to preparation and administration to avoid dosing errors.
VYXEOS is contraindicated in patients with a history of serious hypersensitivity reactions to cytarabine, daunorubicin, or any component of the formulation.
Serious or fatal hemorrhage events, including fatal CNS hemorrhages, associated with prolonged thrombocytopenia, have occurred with VYXEOS. The overall incidence (grade 1-5) of hemorrhagic events was 74% in the VYXEOS arm and 56% in the control arm. The most frequently reported hemorrhagic event was epistaxis (36% in VYXEOS arm and 18% in control arm). Grade 3 or greater events occurred in 12% of VYXEOS-treated patients and in 8% of patients in the control arm. Fatal treatment-emergent CNS hemorrhage not in the setting of progressive disease occurred in 2% of patients in the VYXEOS arm and in 0.7% of patients in the control arm. Monitor blood counts regularly and administer platelet transfusion support as required.
VYXEOS contains daunorubicin, which has a known risk of cardiotoxicity. This risk may be increased in patients with prior anthracycline therapy, preexisting cardiac disease, previous radiotherapy to the mediastinum, or concomitant use of cardiotoxic drugs. Assess cardiac function prior to VYXEOS treatment and repeat prior to consolidation and as clinically required. Discontinue VYXEOS in patients with impaired cardiac function unless the benefit of initiating or continuing treatment outweighs the risk. VYXEOS is not recommended in patients with cardiac function that is less than normal.
Total cumulative doses of non-liposomal daunorubicin greater than
550 mg/m2 have been associated with an increased incidence of
drug-induced congestive heart failure. The tolerable limit appears
lower
(400 mg/m2) in patients who received radiation therapy to the
mediastinum. Calculate the lifetime cumulative anthracycline
exposure prior to each cycle of VYXEOS. VYXEOS is not recommended in
patients whose lifetime anthracycline exposure has reached the
maximum cumulative limit.
Serious or fatal hypersensitivity reactions, including anaphylactic reactions, have been reported with daunorubicin and cytarabine. Monitor patients for hypersensitivity reactions. If a mild or moderate hypersensitivity reaction occurs, interrupt or slow the rate of infusion with VYXEOS and manage symptoms. If a severe or life-threatening hypersensitivity reaction occurs, discontinue VYXEOS permanently, treat the symptoms, and monitor until symptoms resolve.
VYXEOS contains copper. Consult with a hepatologist and nephrologist with expertise in managing acute copper toxicity in patients with Wilson’s disease treated with VYXEOS. Monitor total serum copper, serum non-ceruloplasmin-bound copper, 24-hour urine copper levels, and serial neuropsychological examinations during VYXEOS treatment in patients with Wilson’s disease or other copper-related metabolic disorders. Use only if the benefits outweigh the risks. Discontinue in patients with signs or symptoms of acute copper toxicity.
Daunorubicin has been associated with severe local tissue necrosis at the site of drug extravasation. Administer VYXEOS by the intravenous route only. Confirm patency of intravenous access before administration. Do not administer by intramuscular or subcutaneous route.
VYXEOS can cause embryo-fetal harm when administered to a pregnant woman. Patients should avoid becoming pregnant while taking VYXEOS. If VYXEOS is used during pregnancy or if the patient becomes pregnant while taking VYXEOS, apprise the patient of the potential risk to a fetus. Advise females and males of reproductive potential to use effective contraception during treatment and for 6 months following the last dose of VYXEOS.
The most common adverse reactions (incidence ≥25%) are hemorrhagic events (74%), febrile neutropenia (70%), rash (56%), edema (55%), nausea (49%), mucositis (48%), diarrhea (48%), constipation (42%), musculoskeletal pain (43%), fatigue (39%), abdominal pain (36%), dyspnea (36%), headache (35%), cough (35%), decreased appetite (33%), arrhythmia (31%), pneumonia (31%), bacteremia (29%), chills (27%), sleep disorders (26%), and vomiting (25%).
Please see full Prescribing Information, including BOXED Warning.
References: 1. Lancet JE, Uy GL, Cortes JE, et al. CPX-351 (cytarabine and daunorubicin) liposome for injection versus conventional cytarabine plus daunorubicin in older patients with newly diagnosed secondary acute myeloid leukemia. J Clin Oncol. 2018;36(26):2684-2692.
2. VYXEOS [package insert]. Palo Alto, CA: Jazz Pharmaceuticals.
3. Mitchell JD, Pfeiffer M, Boehmer J, et al. P516: Cardiotoxicity of CPX-351 vs 7+3 in patients with untreated high-risk acute myeloid leukemia. Poster presented at: EHA2023 (European Hematology Association) Hybrid Congress; June 8–11, 2023; Frankfurt, Germany.
4. Mitchell JD, Pfeiffer M, Boehmer J, et al. P516: Cardiotoxicity of CPX-351 vs 7+3 in patients with untreated high-risk acute myeloid leukemia. Hemasphere. 2023;7(Suppl):e178806c.
5. Lalayanni C, Gavriilaki E, Athanasiadou A, et al. Secondary acute myeloid leukemia (sAML): similarly dismal outcomes of AML after an antecedent hematologic disorder and therapy related AML. Clin Lymphoma Myeloma Leuk. 2022;22(4):e233-e240.
6. Capelli D, Menotti D, Florentini A, Sarcani F, Olivieri A. Chapter 7: Secondary acute myeloid leukemia: pathogenesis and treatment. Accessed March 21, 2024. https://www.ncbi.nlm.nih.gov /books /NBK586211 /?report=printable